 Thus, water is H2O and carbon dioxide is CO2. By convention, no subscript is written when a molecule contains only one atom of an element. 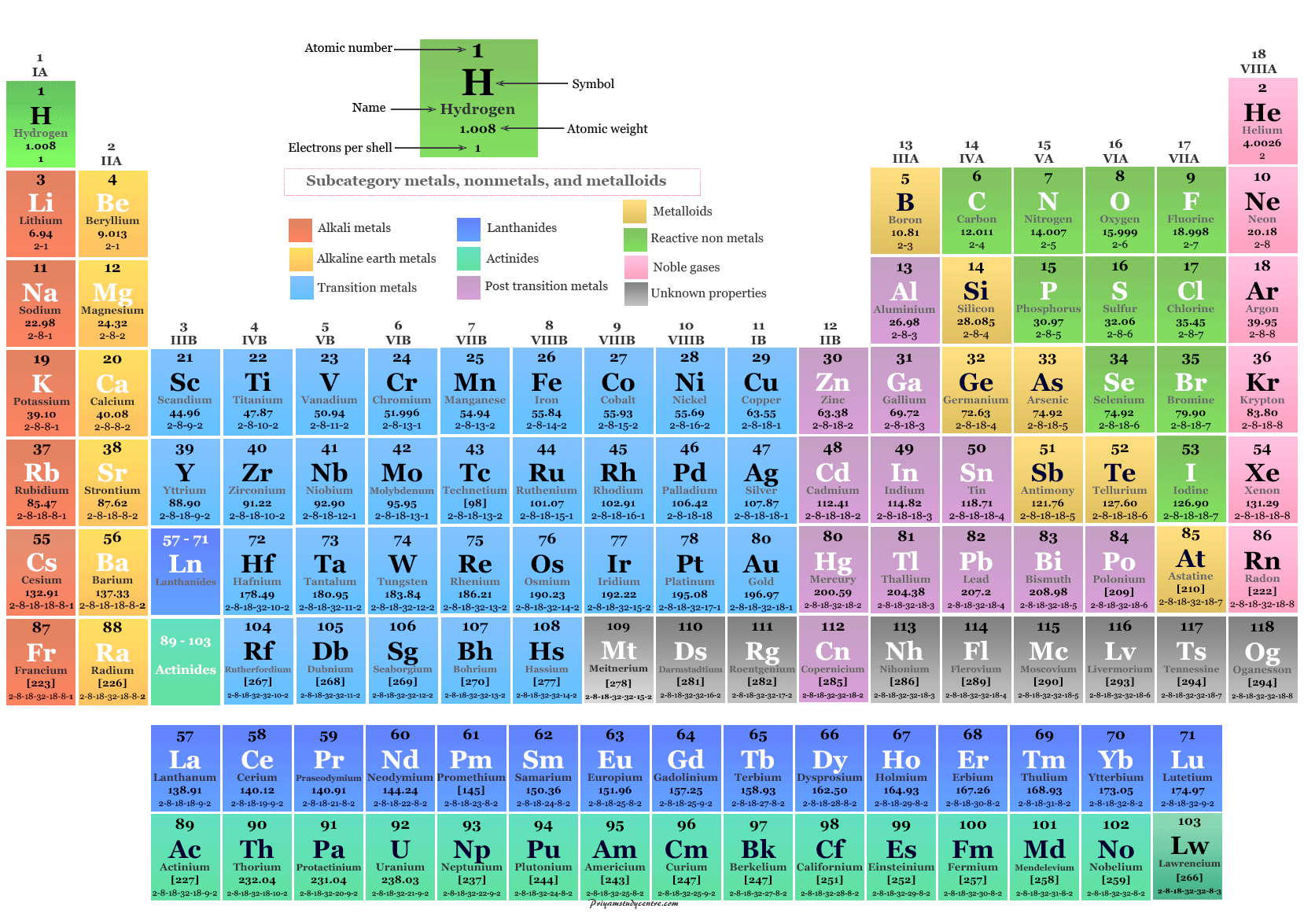
The shorthand notation for a compound describes the number of atoms of each element, which is indicated by a subscript written after the symbol for the element. Nonmetals combine with each other to form covalent compounds, which exist as neutral molecules. These compounds are composed of positive and negative ions formed by adding or subtracting electrons from neutral atoms and molecules. Metals often react with nonmetals to form ionic compounds. When a compound decomposes, the atoms are recovered unchanged Compounds Elements combine to form chemical compounds that are often divided into two categories. Atoms of different elements combine in simple whole numbers to form compounds. Atoms of different elements have different weights and different chemical properties. Matter is made up of atoms that are indivisible and indestructible. Atoms Elements are made up of atoms, the smallest particle that has any of the properties of the element.John Dalton, in 1803, proposed a modern theory of the atom based on the following assumptions. The semimetals can be found along the dividing line between the metals and the nonmetals. A handful of nonmetals are clustered in the upper right corner of the periodic table. Most elements are metals, which are found on the left and toward the bottom of the periodic table. The elements can be divided into three categories that have characteristic properties: metals, nonmetals, and semimetals. A nonrestrictive element is one that offers extra information about the subject or an object in a sentence that is not essential to its meaning. The notation for each element can be found on the periodic table of elements. Each element is represented by a unique symbol. They are therefore the elementary, or simplest, chemical substances - elements. 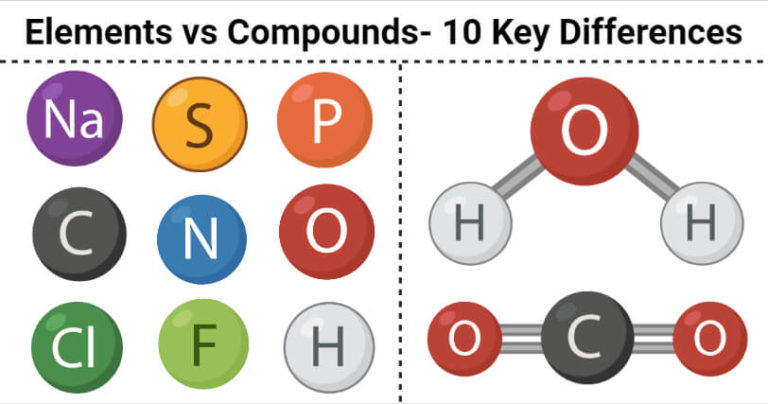
Hydrogen and oxygen, on the other hand, cannot be decomposed into simpler substances. Example: Water decomposes into a mixture of hydrogen and oxygen when an electric current is passed through the liquid. Because atoms cannot be created or destroyed in a chemical reaction, elements such as phosphorus (P4) or sulfur (S8) cannot be broken down into simpler substances by these reactions. It's thought that another one of these magic numbers lies just beyond the range of currently known elements, which means we could potentially discover some new elements that are relatively stable and extremely massive.Any substance that contains only one kind of an atom is known as an element. There are believed to be certain numbers/ratios that are inherently more stable. They help bring imagery and emotion to poetry, stories, and dramas. Archeologists have shown that our ancestors used many of these elements within their ancient oral story. Hence, larger atoms become more unstable. The definition of 'elements of poetry' is 'a set of instruments used to create a poem.' The usages of many of these elements began thousands of years ago. The ratio of neutrons to protons gradually increases to try to compensate, but this results in the neutrons getting more unstable themselves, resulting in more opportunity for weak forces to destabilize them. 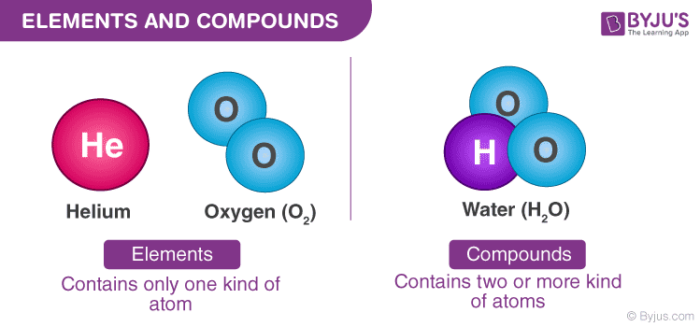
With all of that to consider, as you get larger and larger nuclei with more protons, the electromagnetic forces increase, as well as the distances in the nuclei, putting more strain on the nuclear forces holding it all together. Neutrons will help add to this nuclear force, but they tend to become unstable when they get into too great of concentration and the weak force will cause them to decay into protons. Fusion allows these protons to get close enough together for the stronger, yet short ranged, nuclear force to overpower the electromagnetic force. An element is defined as substances that are cannot be decomposed by a simple chemical process into two or more different materials. The electromagnetic force of the protons wants to push them away from each other. There are several forces at work inside the nuclei of atoms. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
